Will we ever have a cure for the novel coronavirus? That is on the minds of most people around the world right now, ranging from individuals wondering whether it will ever be safe for them to go back to work to business owners wondering how to reopen safely to government officials working tirelessly to stop the spread. There are a lot of obstacles any cure or vaccine will need to clear before being available to the public, but a vaccine might come sooner than you think.
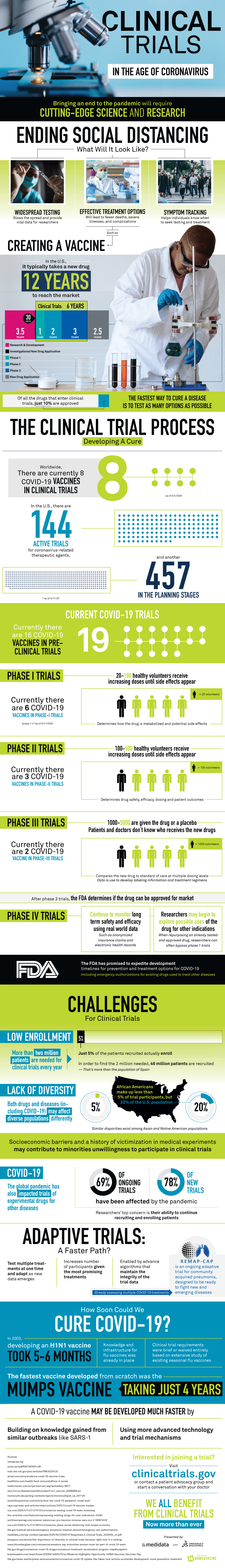
The average time it takes for a new drug to make it to the marketplace is 12 years. That encompasses the research phase, the applications, and the clinical trials. Clinical trials are one of the biggest hold ups because they require many phases with upwards of thousands of participants.
Even if regulators pull out the stops and researchers go full force into clinical trials, the biggest hold up then becomes finding not just participants for the clinical trials, but participants in different genders and ethnicities. People benefit from generations of medical research without thinking about the human sacrifice that made it all possible. In short, if you want a vaccine, participate in a clinical trial.
Currently there are eight vaccines worldwide in clinical trials. In the United States there are 144 active trials for therapeutic agents to use to treat or cure the novel coronavirus, and there are nearly 500 in the planning stages.
The vaccines in the clinical trials show a lot of promise. Two of the vaccines are already in the third stage of trials, which means that if they are proven safe and effective there could be a vaccine on the market by fall or winter.
Still, once the vaccine is approved and proven it will take some time to manufacture it. But there may be an end in sight. Learn more about how clinical trials work from the graphic below.








Reply